Instructions for the Bioviva Wound Dressing product. Bioviva Dressing Soluble Dental Gauze Dressing is intended for use as a wound dressing in extraction sites, and may be used as a wound dressing for the temporary management of oral surgical wounds.
Below are instructions for the Bioviva Wound Dressing product. Bioviva Soluble Dental Gauze Dressing is intended for use as a wound dressing in extraction sites, and may be used as a wound dressing for the temporary management of oral surgical wounds, such as operative, post-operative and traumatic injuries.
Instructions for Use
- Cut or fold Bioviva Soluble Dental Gauze Dressing as needed to cover the wounded area.
- Gently apply to the wounded site for product activation.
- Repeat the steps above with another piece of Bioviva Soluble Dental Gauze Dressing as needed.
- Excess dressing material may be removed by rinsing with sterile water or saline solution. If allergy occurs, stop using bioviva soluble dressing dental gauze dressing or contact a doctor or emergency room.
For additional usage tips, see the case below.
Storage
Bioviva Soluble Dental Gauze Dressing is available sterile - packed individually in different sizes. Keep Bioviva Soluble Dental Gauze Dressing and a pool dry place for maximum shelf life. Preferred storage temperature is between 5°C to 40°C (41°F to 104°F)
Contraindications
Patients with allergies to sodium carboxymethyl cellulose (CMC) should refrain from using this product.
Caution
Federal law restricts this device to sale by or on the order of a physician. Read the instructions before use. Ensure gloves and devices are kept dry before use. The contents of each blister cavity are sterile unless opened or damaged. Discard any unused portion immediately after use. The dressing should not be used on dirty, contaminated or infected extraction sockets. Consult your doctor if you notice any itching, burning, rash or signs of infection such as fever, pain, redness or swelling. This product is not reusable. It is a one-time use.
Case Demonstration
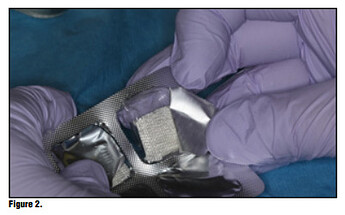
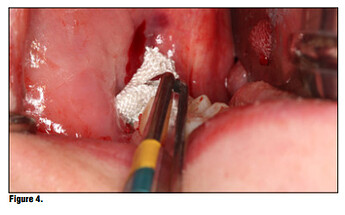
Bioviva is individually packaged. (Figure 1). The product is packed in individual blister packs in 1.9 cm squares for ease of use and to avoid any cross contamination issues (Figure 2). Simply puncture the packaging backing and remove the gauze from the packaging using dry forceps to avoid the gauze from sticking to the forceps (Figure 3). The product can be cut to size if the gauze is larger than the site, or the product can be folded to properly fit using tissue forceps (Figure 4).
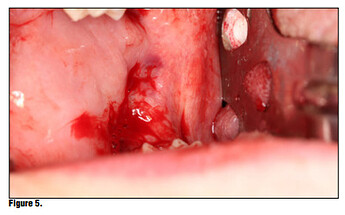
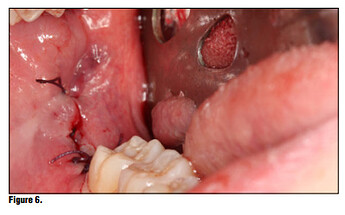
Once the product is removed from the packaging, simply place it onto the wound or extraction site dry and with a matter of 15-20 seconds the material absorbs the wound fluids and turns into a gel and fills the void (Figure 5). Once the site is properly sealed with the material it can be properly rinsed and should not become dislodged. In this case, a suture was placed in conjunction with the product, but in many cases this may not be necessary. A practitioner’s judgment should be utilized in determining if a suture is required in conjunction with the product (Figure 6). The site itself can be sutured, but is not necessary. Nevertheless, the site should not be sutured in place using primary closure as the product needs to be in contact with oral environment and have proper drainage. It is not recommend to suture with primary closure as the creation of a closed contaminated space could be created and this may increase the risk for abscess formation.
File Downloads
Click on the links below to download the files associated with Bioviva.